MetallurgyHard
Question
The chemical process in the production of steel from haematit ore involve :
Options
A.reduction
B.oxidation
C.reduction followed by oxidation
D.oxidation followed by reduction
Solution
The FeO present in haematite ore or obtained during roasting is first oxidised to Fe2O3 and Fe2O3 is reduced finally to Fe.
2FeO + O2
O2  Fe2O3 (osidation during roasting)
Fe2O3 (osidation during roasting)
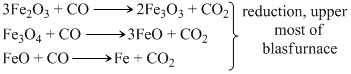
Reduction, upper most part of blast furnace.
2FeO +
 O2
O2  Fe2O3 (osidation during roasting)
Fe2O3 (osidation during roasting)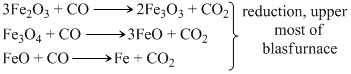
Reduction, upper most part of blast furnace.
Create a free account to view solution
View Solution FreeMore Metallurgy Questions
The process of zone refining is used for :...Match the ores listed in column I with the type of ores listed in column II and select the correct. alternate. Column I ...The iron obtained from the blast furnace is called :...In the extraction of nickel by Mond process, the metal is obtained by:...Which one of the following metals cannot be extracted by carbon reduction ?...