Periodic TableHard
Question
Which has most stable +2 oxidation state ?
Options
A.Sn
B.Pb
C.Fe
D.Ag
Solution
Among the four choices given. Ag and Fe are not correct, as they are not much stable in +2 ox. state.
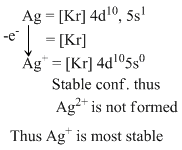

Sn and pb both shows stable +2 oxidation state but to inert effect Pb2+ is more stable than Sn2+
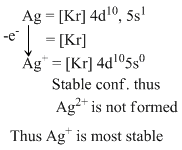

Sn and pb both shows stable +2 oxidation state but to inert effect Pb2+ is more stable than Sn2+
Create a free account to view solution
View Solution FreeMore Periodic Table Questions
Identify the wrong statement in the following:...Beryllium and aluminium exhibit many properties which are similar. But the two elements differ in...Which of the following shows false order :-...Which one of the following does not correctly represent the correct order of the property indicated against it?...In which of the following arrangements the order is NOT according to the property indicated against it?...