Atomic StructureHard
Question
The I.P. of an atom is 36 eV. What is the value of second excitation potential :-
Options
A.5 eV
B.12 eV
C.32 eV
D.27 eV
Solution
Second excitation potential means energy required to move from 1st to 3rd orbit
E1 = - 36 eV
E3 =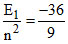 = - 4 eV
= - 4 eV
E3 - E1 = - 4 - (- 36 eV) = 32 eV
E1 = - 36 eV
E3 =
E3 - E1 = - 4 - (- 36 eV) = 32 eV
Create a free account to view solution
View Solution FreeMore Atomic Structure Questions
Correct set of forur quantum numbers for the valence (outermost) electron of rubidium (Z = 37) is :...Select the correct expression(s) according to Bohr model -...The outermost configuration of most electronegative element is :...Rutherford′s experiment on scattering of α- particles showed for the first time that the atom has...A particle X moving with a certain velocity has a debroglie wave length of 1(Ao), If particle Y has a mass of 25% that o...