Boron and Carbon FamilyHard
Question
Hydrocarbon (A) of molecular formula C4H6 gives white ppt with Tollen′s reagent. (B) is the isomer of (A). One mole of (B) picks up one mole of bromine to form 1,4-dibromo-2-butene. Thus (A) and (B) are :-
Options
A.CH3 - C ≡ C - CH3 and CH3 - CH = C = CH2
B.CH3 - CH2 - C ≡ CH and 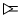 CH2
CH2
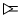 CH2
CH2C.CH3 - C ≡ C - CH3 and 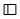
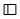
D.CH3 - CH2 - C ≡ CH and CH2 = CH - CH = CH2
More Boron and Carbon Family Questions
In which of the following compounds, then C - Cl bond ionisation shall give most stable carbonium ion?...Which of the following reaction(s) will have final product as hydrocarbon -...The acceptable level of carbon monoxide in the atmosphere in ppm level is :-...B(OH)3 + NaOH ⇋ NaBO2 + Na[B(OH)4] + H2O How can this reaction is made to proceed in forward direction?...Proceeding from the enthalpy of formation of gaseous CO2(ᐃHfo = - 393.5 kJ mol-1) and the thermochemical equation ...