Chemistry miscellaneousHard
Question
The difference between ᐃH and ᐃE at constant volume equal to :-
Options
A.R
B.PᐃV
C.VᐃP
D.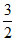 R
R
Solution
ᐃH = ᐃE + PᐃV + VᐃP
ᐃH = ᐃE + VᐃP (at constant volume)
ᐃH - ᐃE = VᐃP
ᐃH = ᐃE + VᐃP (at constant volume)
ᐃH - ᐃE = VᐃP
Create a free account to view solution
View Solution FreeMore Chemistry miscellaneous Questions
Which of the following reaction is incorrect :...Which of the following combination will produce H2 gas?...Cl can be oxidised by :-...The de Broglie wavelength of a tennis ball of mass 60 g moving with a velocity of 10 metres per second is approximately...Ag+ + NH3 ⇋ [Ag(NH3)+] ; k1 = 3.5 × 10-3 [Ag(NH3)]+ + NH3 ⇋ [Ag(NH3)2]+ ; k2 = 1.7 × 10-3 then the...