Carboxylic Acid and Acid DerivativesHard
Question
What weight of Na2CO3 of 95% purity is required to neutralize 50 mL of 0.2 N acid :-
Options
A.0.457 g
B.0.557 g
C.0.53 g
D.0.6572 g
Solution
Milli equivalent of Na2CO3 = milli equivalent of acid
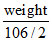 × 1000 = 50 × 0.2
× 1000 = 50 × 0.2
weight =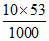 = 0.53 g
= 0.53 g
weight of sample = 0.53 ×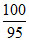 = 0.557g
= 0.557g
weight =
weight of sample = 0.53 ×
Create a free account to view solution
View Solution FreeTopic: Carboxylic Acid and Acid Derivatives·Practice all Carboxylic Acid and Acid Derivatives questions
More Carboxylic Acid and Acid Derivatives Questions
Which is the correct statement in regards to CH3COCl -...In the folowing reaction,The major product is-...In the presence of small amount of phosphorous, aliphatic carboxylic acids react with chlorine or bromine to yield a com...In which reaction product is hydrocarbon?...In the given reaction :[X] + Acetic anhydride → Aspirin [X] will be:...