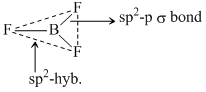Chemical BondingHard
Question
The geometry and the type of hybrid orbital present about the central atom in BF3 is :
Options
A.linear, sp
B.trigonal planar, sp2
C.tetrahedral, sp3
D.pyramidal, sp3
More Chemical Bonding Questions
The Cl - Cl - Cl angle in 1, 1, 2, 2-tetracholroethene and tetrachloromethane respectively will be about :...The shape of NH3 molecule is :...In which of the following pairs of molecules/ ions, the central atoms have sp2 hybridisation ?...For the cell given below ECell - Eocell = - 0.118 V Zn/Zn2+ || Cu2+/CuWhat is the ratio of [Zn2+]/[Cu+2] :-...The helical structure of proteins is stabilised by:-...