Chemical BondingHard
Question
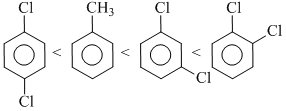
Arrange the following compounds in order of increeasing dipole moment toluene (I) m -dichlorobenzene (II), o-dichlorobenzene (III), p-dichlorobenzene (IV) :
Options
A.I < IV < II < III
B.IV < I < II < III
C.IV < I < III < II
D.IV < II < I < III
More Chemical Bonding Questions
At least one bond pair is situated exactly opposite to another bond pair in -...Which of the following best explains the reason for the relative stabilities of the conformers shown?...After understanding the assertion and reason, choose the correct option.Assertion : In the bonding molecular orbital (MO...Among the following, the linear molecule is :...When the value of (n + l) is not more than 3 which of the following subshells is not possible to exist :-...