ThermochemistryHard
Question
Consider the following reactions :
C (s) + O2 (g) → CO2 (g) + x kJ
CO (g) +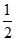 O2 (g) → CO2 (g) + y kJ
O2 (g) → CO2 (g) + y kJ
The heat of formation of CO (g) is :
C (s) + O2 (g) → CO2 (g) + x kJ
CO (g) +
The heat of formation of CO (g) is :
Options
A.- (x + y) kJ / mol
B.(x - y) kJ / mol
C.(y - x) kJ / mol
D.None of these
Solution
C(s) + O2(g) → CO2(g);
ᐃrH1 = - x kJ/mol .....(1)
CO(g) + 1/2O2(g) → CO2(g)
ᐃH2 = - y kJ/mole so, C(s) +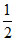 O2 (g) → C(g) ᐃH = y - x
O2 (g) → C(g) ᐃH = y - x
ᐃrH1 = - x kJ/mol .....(1)
CO(g) + 1/2O2(g) → CO2(g)
ᐃH2 = - y kJ/mole so, C(s) +
Create a free account to view solution
View Solution FreeMore Thermochemistry Questions
The enthalpy of neutralization of a strong monobasic acid by a strong monoacidic base is –13,700 cal. A certain monobasi...Match the transformations in column I with appropriate options in column II...Study the following thermodynamic data given by E. H. P. Cordfunke, A. S. Booji and M. Y. Furkalionk.(i) DyCl3(s) → DyCl...The thermochemical equation for the dissociation of hydrogen gas into atoms may be written asH2 → 2H; ΔH = 432.0 kJ.What...The standard molar enthalpies of formation of cyclohexane (l) and benzene(l) at 298 K are −156 and +49 kJ/ mol, respecti...