General Organic ChemistryHard
Question
Which of the following resonance structures is the major contributor to the resonance hybrid ?
CH3 − CH2 −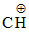 −
− 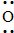 CH3 ⇔ CH3 − CH2 − CH =
CH3 ⇔ CH3 − CH2 − CH = 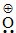 CH3
CH3
CH3 − CH2 −
Options
A.I
B.II
C.Both have equal contribution
D.They are not resonance structures
More General Organic Chemistry Questions
Which of the following statements are correct for nucleophile :...Geometrical isomerism is possible in case of :...Which one of the following is the most acidic?...Rate of the reaction is fastest when Z is...Give the correct order of decreasing basicity of the following compounds,C6H5NH2 (C6H5)2NH (C6H5)3N C6H11NH2 I II III IV...