Chemical EquilibriumHard
Question
N2O4(g) ⇋ 2NO2(g), Kc = 4. This reversible reaction is studied graphically as shown in figure. Select the correct statements out of I, II and III.
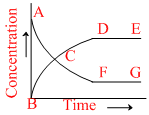
I : Reaction quotient has maximum value at point A
II : Reaction proceeds left to right at a point when
[N2O4] = [NO2] = 0.1 M
III : Kc = Q when point D or F is reached :

I : Reaction quotient has maximum value at point A
II : Reaction proceeds left to right at a point when
[N2O4] = [NO2] = 0.1 M
III : Kc = Q when point D or F is reached :
Options
A.I, II
B.II, III
C.I, III
D.I, II, III
Solution
(I) N2O4 2NO2 ⇋ Kc = 4
at point − A
Q =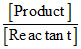 = 0
= 0
So, Q have minimum value at point A.
(II) at point [N2O4] = [No2] = 0.1m
Q =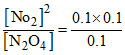
Q < Kc
So, reaction proceeds left to right
(III) Kc = Q at point [D & F].
at point − A
Q =
So, Q have minimum value at point A.
(II) at point [N2O4] = [No2] = 0.1m
Q =
Q < Kc
So, reaction proceeds left to right
(III) Kc = Q at point [D & F].
Create a free account to view solution
View Solution FreeMore Chemical Equilibrium Questions
For the given reaction 2A(s) + B(g) $\rightleftharpoons$C(g) + 2D(s) + E(s), the extent of reaction of B was found to be...Consider the following gaseous equilibrium in a closed container of volume "V" at T(K).$$P_{2}(\text{ }g) + Q_{2}(\text{...The standard emf of cell, involving one electron change is found to be 0.591 V at 25ºC. The equilibrium constant of the ...An aqueous solution of volume 500 ml, when the reaction 2Ag+ (aq) + Cu(s) $\rightleftharpoons$Cu2+ (aq) + 2Ag(s) reached...An amount of 16 moles H2 and 4 moles of N2 is confined in a vessel of volume one litre. The vessel is heated to a consta...