Aromatic HydrocarbonsHard
Question
An organic compound having the molecular formula C8H10O on being heated with I2 and dilute NaOH gives a yellow precipitate. The compound is expected to be -
Options
A.C6H5CH2CH2OH
B.CH3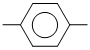 CH2OH
CH2OH
 CH2OH
CH2OHC.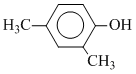

D. CHOHCH3
CHOHCH3
 CHOHCH3
CHOHCH3More Aromatic Hydrocarbons Questions
Amongst the following, the moderately activating group is...The compounds P, Q and S were separately subjected to nitration using HNO3/H2SO4 mixture. The major product formed in ea...Benzene is obtained by all the following reactions except -...The electrophile, E+ attacks the benzene ring to generate the intermediate σ-complex. Of the following, which 	...In Friedel-Crafts acylation reaction the electrophile is -...