Atomic StructureHard
Question
Electromagnetic radiation with maximum wavelength is :
Options
A.ultra-violet
B.radio- wave
C.X-ray
D.infra-red
Solution
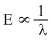
thus smaller is the energy , larger is the wavelength.
Among the given radiations, radio-waves are least energtic.
Radiaation UV Radio -waves
λ(cm) 10-6 - 10-5 1 - 102
X-rays IR
≥ 10-6 10-4 to 10-3
Create a free account to view solution
View Solution FreeMore Atomic Structure Questions
The number of electrons having m = 0 for sodium atom is...If the nitrogen atom had electronic configuration 1s7, it would have energy lower than that of the normal ground state c...The energy required by electrons, present in the first Bohr orbit of hydrogen atom to be excited to second Bohr orbit is...Which of the following electromagnetic radiation have greater frequency?...The correct set of four quantum numbers for the unpaired electron of chloride atom is...