SolutionHard
Question
The freezing point of aqueous solution that contains 3% urea, 7.45% KCl and 9% of glucose is (given Kf of water = 1.86 and asume molarity = molality).
Options
A.290 K
B.285.5 K
C.267.42 K
D.250 K
Solution
ᐃTf = i.m. Kf
ᐃTf = i1m1Kf + i2 m2 Kf + i3 m3 Kf = (m1 + 2m2 + m3) Kf
ᐃTf =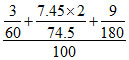 × 1000 × 1.86
× 1000 × 1.86
ᐃTf = 3 × 1.86 = 5.58
Tf of solution = 273 - 5.58 = 267.42 K Ans.
ᐃTf = i1m1Kf + i2 m2 Kf + i3 m3 Kf = (m1 + 2m2 + m3) Kf
ᐃTf =
ᐃTf = 3 × 1.86 = 5.58
Tf of solution = 273 - 5.58 = 267.42 K Ans.
Create a free account to view solution
View Solution FreeMore Solution Questions
Mixture of volatile components A and B has total vapour pressure (in Torr) p = 254 - 119 xA where xA is mole fraction of...A maxima or minima obtained in the temperature composition curve of a mixture of two liquids indicates...1 mol each of 1the following compounds is dissolved in 1L of solution. Which will have the largest ᐃTb value ?...If ′A′ contains 2% NaCl and is separated by a semipermeable membrane from ′B′ which contains 10%...The vapour pressure of a dilute aqueous solution of glucose is 750 mm of mercury at 373 K. The mole fraction of solute i...