SolutionHard
Question
Two elements A and B form compounds having molecular formulae AB2 and AB4. When dissolved in 20.0 g of Benzene, 1.00 g of AB2 lowers f.p. by 3.2oC whereas 1.00 g of AB4 lowers f.p. by 1.3oC. The molal depression constant for benzene is 5.1. The atomic masses of A and B are respectively
Options
A.can′t be predicted
B.42.6, 25.59
C.30.60
D.25.59, 42.6
Solution
ᐃTf = 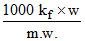
for AB2 = 3.2 =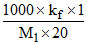
for AB4 = 1.3 =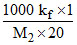
M1 = 110.67, M2 = 196.15
for AB2 = a + 2b = 110.87 ......... (i)
for AB4 = a + 4b = 196.15 ......... (ii)
By solving eq (i) and (ii)
a = 25.59 b = 42.64
for AB2 = 3.2 =
for AB4 = 1.3 =
M1 = 110.67, M2 = 196.15
for AB2 = a + 2b = 110.87 ......... (i)
for AB4 = a + 4b = 196.15 ......... (ii)
By solving eq (i) and (ii)
a = 25.59 b = 42.64
Create a free account to view solution
View Solution FreeMore Solution Questions
The vapour pressure of a dilute aqueous solution of glucose is 750 mm of mercury at 373 K. The mole fraction of solute i...Freezing point lowering expression is ᐃTf = Kfm (molality)Which of the following assumptions are considered for th...FeCl3 on reaction with K4 [Fe(CN)6] in aq. solution gives blue colour. These are separated by a semipermeable membrane P...Which of the following is less than zero for ideal solutions ?...Which one of the following statements is correct when SO2 is passed through acidified K2Cr2O7 solution?...