Gaseous StateHard
Question
When an ideal gas undergoes unrestrained expansion, no cooling occure because the molecules :
Options
A.are above the inversion temperature
B.exert no attractive forces on each other
C.do work equal to loss in kinetic energy
D.collide without loss of energy
Solution
Because there is no intermolecular force of attraction between gas molecules.
P′ = P × mole fraction or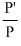 = mole fraction
= mole fraction
P′ = P × mole fraction or
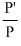 = mole fraction
= mole fractionCreate a free account to view solution
View Solution FreeMore Gaseous State Questions
The rates of diffusion of SO3, CO2, PCl3 and SO2 are in the following order -...Henry′s law constants for four gases are given below at 293 K. Which one is more soluble in water at that temperat...A thin balloon filled with air at 47oC has a volume of 3 litre. If on placing it in a cooled room its volume becomes 2.7...A and B are two identical vessels. A contains 15 g ethane at 1atm and 298 K. The vessel B contains 75 g of a gas X2 at s...The order of ionisation potential between He+ ion and H-atom (both species are in gaseous state) is :-...