Nitrogen Containing CompoundHard
Question
When nitrobenzene is treated with Br2 in presence of FeBr2, the major product formed is m-bromonitro-benzene. Statements which are related to the formation m-isomer, are :
Options
A.The electron density on meta- carbon is more than that on ortho and - positions
B.The intermediate carbonium ion formed after initial attack of Br+ at the meta-position is least destablished
C.Loss of aromaticity when Br+ attacks at the ortho and para -positions and not at meta - position
D.Easier loss of H+ to regain aromaticity from the, eta-position than from ortho and para-positions
Solution
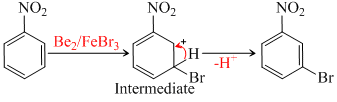
Attack of E+ ( - Br) on m-position proves that it has higher electron degain. Intermediate removes - H+ easily to regain its aromatic character.
Create a free account to view solution
View Solution FreeMore Nitrogen Containing Compound Questions
Which of the following would undergo Hoffmann bromide reaction to form primary amine-...Propylamine reacts with nitrous acid to from a relatively stable cation viz. -...R′ − N is a-...'A' is a neutral organic compound (M. F : $C_{8}H_{9}ON$ ). On treatment with aqueous ${Br}_{2}/{HO}^{( - )}$, 'A' forms...C6H5NH2 + CS2 C6H5NCS = HgS + 2HCl the above reaction is :...