Carboxylic Acid and Acid DerivativesHard
Question
(A), (B) and (C) are three dicarboxylic acids such that -
(a)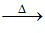 carboxylic acid + CO2 ↑
carboxylic acid + CO2 ↑
(b)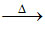 acid anhydride + H2O ↑
acid anhydride + H2O ↑
(c)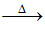 cyclic ketone + H2O + CO2 ↑
cyclic ketone + H2O + CO2 ↑
(a)
(b)
(c)
Options
A.(a) → adipic acid
(b) → malonic acid
(c) → succinic acid
(b) → malonic acid
(c) → succinic acid
B.(a) → Malonic acid
(b) → adipic acid
(c) → succinic acid
(b) → adipic acid
(c) → succinic acid
C.(a) → adipic acid
(b) → succinic acid
(c) → malonic acid
(b) → succinic acid
(c) → malonic acid
D.(a) → Malonic acid
(b) → succinic acid
(c) → adipic acid
(b) → succinic acid
(c) → adipic acid
Topic: Carboxylic Acid and Acid Derivatives·Practice all Carboxylic Acid and Acid Derivatives questions
More Carboxylic Acid and Acid Derivatives Questions
In the molecule CH3CHCl − CH2COOH, Cl atom is attached to -...Which of the following dicarboxylic acids contains the most acidic hydrogen ?...When acetyl chloride reacts with Grignard′s reagent we get -...Sodium salt of which one of the monobasic acids on electrolysis does not give hydrocarbon:...Which of the following statement is not applicable to H2SO4 :-...