Carbonyl CompoundHard
Question
Compound A (molecular formula C2H8O) is treated with acidified potassium dichromate to form a product B (molecular formula C2H6O). B forms a shining silver mirror on warming with an aqueous solution of H2NCONHMH2 and sodium acetate gives a product C. Identify the structure of C.
Options
A.CH3CH2CH = N.NHCOH2
B.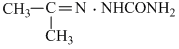
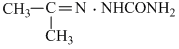
C.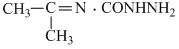
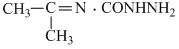
D.CH3CH2CH = N.CONHNH2
Solution
A is p-alcohol because its oxidation gives an aldehyde ′B′. B gives shining mirror with ammonical silver nitrate (Tollen′s reagent)
Thus structure of compound A is CH3Ch2CH2OH(M.F. = C3H8O)
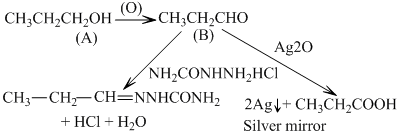
Thus structure of compound A is CH3Ch2CH2OH(M.F. = C3H8O)

Create a free account to view solution
View Solution Free