Carbonyl CompoundHard
Question
Which of the following has the most acidic hydrogen ?
Options
A.3-hexanone
B.2, 4-hexadione
C.2, 3-hexadione
D.2, 3-hexadione
Solution
(a) 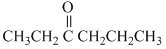
(b)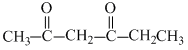
(c)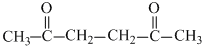
(d)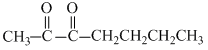
In (b) : - CH2 - gp. (methylene group) is sandwitched between two electron withdrawing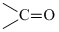 gps. thus its H-atoms are most acidic.
gps. thus its H-atoms are most acidic.
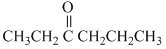
(b)
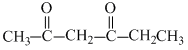
(c)
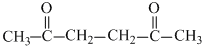
(d)
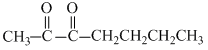
In (b) : - CH2 - gp. (methylene group) is sandwitched between two electron withdrawing
 gps. thus its H-atoms are most acidic.
gps. thus its H-atoms are most acidic.Create a free account to view solution
View Solution FreeMore Carbonyl Compound Questions
Acetone is treated with excess of ethanol in the presence of hydrochloric acid. The product obtained is:...Acetaldehyde cannot give...In the Cannizzaro reaction 2PhCHO PhCH2OH + PhCOO− the slowest step is...In the Cannizaro′s reaction given below : 2Ph - CHO → Ph - CH2OH + PhCO2- The slowest step is:...Which of the following statements is wrong:...