Alcohol, Phenol and EtherHard
Question
In the folowing compounds
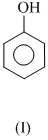
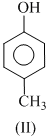
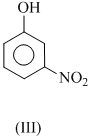
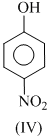
The order of acidity is :




The order of acidity is :
Options
A.III > VI > I > II
B.II > I > II > IV
C.II > I > III > IV
D.IV > III > I > II
Solution




(i) All the four acidic nature due to heterolysis of phenoli - O - H group.
(ii) Any electron releasing group will secreases the acidic strength, the (II) will be less acidic than phenol.
(iii) - NO2 gp . is rlrctron with drawing and so increases the acidic strength. Electron pulling effect is more at p-position than -position. That why p-nitrophenol (IV) is most acidic. Thus, order of acidic strength is IV > III > I > II
Create a free account to view solution
View Solution FreeMore Alcohol, Phenol and Ether Questions
Unacceptable name for a compound containing one -OH group attached to benzene nucleus would be-...Chlorination of roluene in the presence of light and heat followed by treatment with aqueous NaOH gives :...The electrophile involved in the above reaction is...The correct sequence of reagents for the above conversion of X to Y is :...n-propyl bromide on treatment with ethanolic postassium hydroxide produces :...