Alcohol, Phenol and EtherHard
Question
1-Chloro-2-butene is allowed to react with a concentrated solution of C2H5ONa in C2H5OH The rate of reaction is found to be dependent on the concentrations of both the substrate and the nucleophile.
CH3CH = CHCH2Cl + C2H5ONa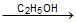
The major product formed in the reaction is
CH3CH = CHCH2Cl + C2H5ONa
The major product formed in the reaction is
Options
A.CH3CH = CHCH2OC2H5
B.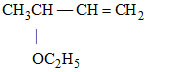
C.CH2 = CH − CH = CH2
D.All of these in equal amounts