Alcohol, Phenol and EtherHard
Question
HBr reacts fastest with :
Options
A.2- methyl propan -2-ol
B.propan-1-ol
C.propan-2-ol
D.2- methyl propan -1-ol
Solution
The reaction takes place as follows:
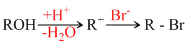
Thus an alcohol which gives more stable carbocation will be moer reactive in this reaction. 2-methyl propan -2-ol (a) will give most stable t-carbocation among these most readily. (b) and (d) will give p-carbocation which (c) gives s-carbocation.
Thus an alcohol which gives more stable carbocation will be moer reactive in this reaction. 2-methyl propan -2-ol (a) will give most stable t-carbocation among these most readily. (b) and (d) will give p-carbocation which (c) gives s-carbocation.
Create a free account to view solution
View Solution FreeMore Alcohol, Phenol and Ether Questions
First member of ether series on reaction with sodium and liquified ammonia gives -...Mustard oil reaction will be given by :-...The product of the reaction of benzene with oxygen in the presence of V2O5 as catalyst at 200oC is -...In the reaction CH3CH = CH2 A Bthe product (B) is -...Phenol is more acidic than cyclohexanol because -...