JEE AdvancedCoordination CompoundHard
Question
Consider the following statements :
S1 : Generally square planar complexes show geometrical isomerism but do not exhibit optical isomerism because they do not possess plane of symmetry.
S2 : ᐃt =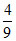 ᐃ0
ᐃ0
S3 : In octahedral complexes each electron entering the t2g orbitals stabilizes the complex ion by 0.4 ᐃo and each electron entering the eg orbital destabilizes the complex by an amount of 0.6 ᐃo.
Select the correct statement from the codes given below
S1 : Generally square planar complexes show geometrical isomerism but do not exhibit optical isomerism because they do not possess plane of symmetry.
S2 : ᐃt =
S3 : In octahedral complexes each electron entering the t2g orbitals stabilizes the complex ion by 0.4 ᐃo and each electron entering the eg orbital destabilizes the complex by an amount of 0.6 ᐃo.
Select the correct statement from the codes given below
Options
A.S1 and S3 are correct
B.S2 and S3 are correct
C.S1 is incorrect
D.S2 and S3 are incorrect
Solution
S1 : Square planar complexes with symmetrical ligands like (en) do not show geometrical isomerism and they have plane of symmetry, so optically inactive.
S2 and S3 are correct statements.
S2 and S3 are correct statements.
Create a free account to view solution
View Solution FreeMore Coordination Compound Questions
Which of the following is not onsidered as an organometallic compound ?...CuSO4 and KCN react to produce :...In the complex FeK2[Fe(CN)6],...In the isoelectronic series of metal carbonyl, the C–O bond strength is expected to increase in the order....Which of the following is not considered as an organometallic compound?...