Alkyl Halides and Aryl HalidesHard
Question
In the preparation of ethyl chloride by the reaction CH3CH2OH + HCl 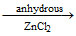 CH3CH2Cl + H2O
CH3CH2Cl + H2O
the major reason for using anhydrous ZnCl2 as a catalyst is that
the major reason for using anhydrous ZnCl2 as a catalyst is that
Options
A.It absorbs water formed in the reaction and drives the equilibrium to the right
B.It coordinates with the product CH3CH2Cl and drives the reaction to the right
C.It assists in forming the nucleophile Cl− from HCl
D.It coordinates with oxygen of ethyl alcohol and increases the leaving group ability of the−OH group
More Alkyl Halides and Aryl Halides Questions
(A) (B) (C) Product A, B & C are -...Chloroform can be obtained from -...Steps I and II are...The order of decreasing SN1 reactivities of the halides CH3CH2CH2Cl CH2 = CHCHClCH3 CH3CH2CHClCH3 I II III is -...Correct order of reactivity of following acid derivatives is MeCOCl MeCON3 MeCOOCOMe I II III[This order is only for nuc...