Alkyl Halides and Aryl HalidesHard
Question
Which of the following reactions is not expected to give a satisfactory yield of alkyl iodide -
Options
A.CH3CH2OH + KI + H3PO4 →
B.CH3CH2OH + Pl3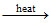
C.CH3CH2Br + Nal 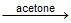
D.CH3CH2COOAg + I2 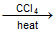
More Alkyl Halides and Aryl Halides Questions
What are A & B in the following reaction?...The product P of the following reaction is Product (P) Major...An aromatic primary amine (A) is heated with another compound (B) in the presence of alcoholic KOH to give a bad-smellin...Which of the following leads to the formation of an alkyl halide -...Consider the following sequence of reactions.C2H5Cl X YThe products (X) and (Y) are, respectively -...