Coordination CompoundHardBloom L2
Question
Which of the following ions are optically active?
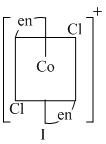
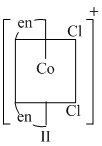
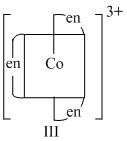
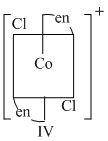




Options
A.I only
B.II only
C.II and III
D.IV only
Solution
(B) II is cis form which lacks centre of symmetry as well as plane of symmetry ; so optically active
(C) (II) and (III) lack symmetry elements ; so both are optically active.
(D) IV is trans form which has centre of symmetry as well as plane of symmetry ; so optically inactive
Create a free account to view solution
View Solution FreeMore Coordination Compound Questions
Which one of the following has the regular tetrahedral structure?...Which one of the following complexes in an outer orbital complex ?...[Co(NH3)5NO2]Cl2 and [Co(NH3)5ONO]Cl2 are related to each other as :...On treatment of [Pt(NH3)4]2+ with concentrated HCl, two compounds (I) and (II) having the same formula, [Pt(NH3)2Cl2] ar...The ligands in anti cancer drug cis-plantin are :...