Coordination CompoundHard
Question
Arrange the following in order of decreasing number of unpaired electrons?
I : [Fe(H2O)6]2+ II : [Fe(CN)6]3- III : [Fe(CN)6]4- IV : [Fe(H2O)6]3+
I : [Fe(H2O)6]2+ II : [Fe(CN)6]3- III : [Fe(CN)6]4- IV : [Fe(H2O)6]3+
Options
A.IV, I, II, III
B.I, II, III, IV
C.III, II, I, IV
D.II, III, I, IV
Solution
(I) [Fe(H2O)6]2+, 3d6 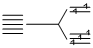 n = 4 (weak field ligand)
n = 4 (weak field ligand)
(II) [Fe(CN)6]3-, 3d5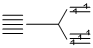 n = 1 (strong field ligand)
n = 1 (strong field ligand)
(III) [Fe(CN)6]4-, 3d6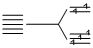 n = 0 (strong field ligand)
n = 0 (strong field ligand)
(IV) [Fe(H2O)6]3-, 3d5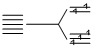 n = 5 (weak field ligand)
n = 5 (weak field ligand)
 n = 4 (weak field ligand)
n = 4 (weak field ligand)(II) [Fe(CN)6]3-, 3d5
 n = 1 (strong field ligand)
n = 1 (strong field ligand) (III) [Fe(CN)6]4-, 3d6
 n = 0 (strong field ligand)
n = 0 (strong field ligand) (IV) [Fe(H2O)6]3-, 3d5
 n = 5 (weak field ligand)
n = 5 (weak field ligand)Create a free account to view solution
View Solution FreeMore Coordination Compound Questions
Which of the following statements about Fe(CO)5 is correct?...Na2S + Na2[Fe(CN)5NO] → Na4[Fe(CN)5NOS] ; oxidation number of Fe in reactant (complex) and product (complex) are :...The oxidation state of Mo in its oxido-complex species [Mo2O4(C2H4)2(H2O)2]2- is...The crystal field stabilization energy (CFSE) is the highest for :...Diethylenetriamine is:...