Coordination CompoundHard
Question
What is the ratio of uncomplexed to complexed Zn2+ ion in a solution that is 10 M in NH3, if the stability constant of [Zn(NH3)4]2+ is 3 × 109 ?
Options
A.3.3 × 10-9
B.3.3 × 10-11
C.3.3 × 10-14
D.3 × 10-13
Solution
Zn2+ + 4NH3 ⇋ [Zn(NH3)4]2+.
Kf =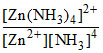 ⇒
⇒ 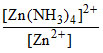 = Kf[NH3]4.
= Kf[NH3]4.
= 3 × 109 × 104 = 3 × 1013.
⇒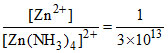 = 3.3 × 10-14.
= 3.3 × 10-14.
Kf =
= 3 × 109 × 104 = 3 × 1013.
⇒
Create a free account to view solution
View Solution FreeMore Coordination Compound Questions
When AgNO3 is added to a solution of Co(NH3)5Cl3, the precipitante of AgCI shows two ionizable chloride ions. This mean:...Given below are two statements:Statement-I Hybridisation, shape and spin only magnetic moment of $K_{3}\left\lbrack Co\l...Which one of the following complexes is an outer orbital complex?...In Ziesses salt C = C bond length is : Note :...Which is correct for product, P, Q and R :-...