Aromatic HydrocarbonsHard
Question
The reaction of toluene with chlorine in presence of ferric chloride gives predominantly :
Options
A.benzoyl chloride
B.m-chlorotoluene
C.benzyl chloride
D.o-and p-chlorotoluene
Solution
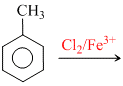
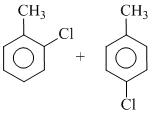
In presence of Fe, E+ substitution takes place in the ring. Because - CH3 gp. is o/p directing, that is why incoming E+(-Cl+) goes at o/p position . If reaction is carried out in pres ence of sun light then substitution takes place in the side chain giving benzyl chloride.
Create a free account to view solution
View Solution FreeMore Aromatic Hydrocarbons Questions
Benzene reacts with benzoyl chloride to form -...The species responsible for nitration and sulphonation by nitric acid, conc. H2SO4 and fuming H2SO4 are -...Nitration of xylene gives only one nitrodimethyl benzene. Structure of xylene is...An aromatic compound ′A′ C7H6Cl2, gives AgCl on bonding with alcoholic AgNO3 solution, and yields C7H7OCl on...When chlorine is passed in cold toluene in the presence of iodine, the product obtianed is -...