HydrocarbonsHard
Question
The nodal plane in the π - bond of ethene is located in :
Options
A.the molecular plane
B.a plane parallel to the molecular plane
C.a plane perpendicular to the molecular plane which bisets the carbon -carbon σ - bond at right angle
D.a plane perpendicular to the molecular plane which contains the carbon-carbon σ - bond
Solution
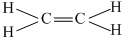 or
or 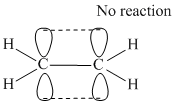
π - bond is formed by lateral overlapping between p-orbitals of C-atoms. These p-obitals are perpendicular to the plane of sp2 hybrid or orbitals or molecular plane .It does not contain any π - electron density and thus nodal plane lies along the molecular plane for π - electron
Create a free account to view solution
View Solution FreeMore Hydrocarbons Questions
HBr reacts with CH2 = CH - OCH3 under anhydrous conditions at room temperature to give...Propene on reaction with diimide (NH = NH, generated in situ by the oxidation of NH2NH2 with H2O2) gives -...Match list I with list II and then select the correct answer from the codes given below the lists.List I A. Isobutane &#...Monochlorination of monodeuterioethane (CH3CH2D) yields -......