HydrocarbonsHard
Question
Consider the following reaction sequence.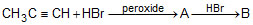 The products (A) and (B) are, respectively,
The products (A) and (B) are, respectively,
Options
A.CH3CH = CHBr and CH3 CHBrCH2Br
B.CH3CBr = CH2 and CH3CBr2 CH3
C.CH3CH = CHBr and CH3CH2CHBr2
D.CH3CH = CHBr and CH3CBr2CH3
More Hydrocarbons Questions
Propene is allowed to react with B2D6 and the product is treated with acetic acid. The final product obtained is -...RCH = CH2 adds on water in the presence of dilute sulphuric acid or phosphoric acid to form -...Acidic hydrogen is present in :...Which of the following hydrocarbon with formula C8H18 gives one monochloro derivatives -...In dehydrohalogenations the base ( alcoholic KOH ) abstracts -...