Coordination CompoundHard
Question
How many moles of AgCl would be obtained, when 100 ml of 0.1 M Co(NH3)5Cl3 is treated with excess of AgNO3?
Options
A.0.01
B.0.02
C.0.03
D.none of these
Solution
The complex is [Co(NH3)5Cl]Cl2 and there are two moles of Cl which can react with Ag+ to form white precipitate of AgCl.
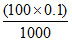 × 2 = 0.02 mole of AgCl
× 2 = 0.02 mole of AgCl
Create a free account to view solution
View Solution FreeMore Coordination Compound Questions
The IUPAC name of Xe [Pt F6] is...The formula of the complex hydridotrimethoxidoborate (III) ion is:...Which molecule/ion among the following cannot act as a ligand in complex compounds?...The compound which does not show paramagnetism ?...Aqueous solution of FeSO4 gives tests for both Fe2+ and SO42– but after addition of excess of KCN, solution ceases to gi...