MetallurgyHard
Question
Roasting of copper pyrites is done :
Options
A.to remove moisture.
B.to oxidise free sulphur and antimony.
C.to convert pyrites completely into Cu2O and FeO.
D.to remove volatile organic impurities.
Solution
(C) is incorrect statement as 2CuFeS2 + 4O2 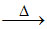 Cu2S + 2FeO + 3SO2.
Cu2S + 2FeO + 3SO2.
Create a free account to view solution
View Solution FreeMore Metallurgy Questions
Addition of high proportions of manganese makes steel useful in making rails of railroads, because manganese...In electrolytic refining of lead, electrolyte is consist of :...Which of the following process is not associated with recovery of the silver -...Which one of the following reactions represents a calcination reaction?...In the metallurgy of iron, the upper layer obtained in the bottom of blast furnace mainly contains :...