MetallurgyHard
Question
Of the following reduction processes, the correct process(es) is/are :
Options
A.Fe2O3 + CO → Fe + CO2
B.ZnO + C → Zn + CO
C.Cu2O + Cu2S → Cu + SO2
D.PbO + C → Pb + CO
Solution
All processes are correct.
(A) Fe2O3 + 4CO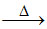 3Fe + 4CO2
3Fe + 4CO2
(B) ZnO + C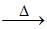 Zn + CO
Zn + CO
(C) 2Cu2O + Cu2S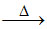 6Cu + SO2
6Cu + SO2
(D) PbO + C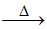 Pb + CO
Pb + CO
(A) Fe2O3 + 4CO
(B) ZnO + C
(C) 2Cu2O + Cu2S
(D) PbO + C
Create a free account to view solution
View Solution FreeMore Metallurgy Questions
Which one of the following metals cannot be extracted by carbon reduction ?...Which process of purification is represented by the following equation :Ti (Impure) + 2I2 $\overset{\quad 250ºC\quad}{\r...Calcium silicate (slag) formed in the slag formation zone in extraction of iron from haematite ore :...Copper and tin are refined by :...Dow's process...