MetallurgyHard
Question
In Van Arkel method, if I2 is introduced at 1800 K over impure zirconium metal, the product will be :
Options
A.iodide of the metal
B.pure metal
C.impurities react with iodine
D.none of these
Solution
Zr (impure) + 2I2 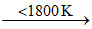 ZrI4 ; ZrI4
ZrI4 ; ZrI4 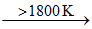 Zr (pure) + 2I2 and hence over 1800oC practically no reaction can take place between Zr and I2.
Zr (pure) + 2I2 and hence over 1800oC practically no reaction can take place between Zr and I2.
Therefore, (D) option is correct.
Therefore, (D) option is correct.
Create a free account to view solution
View Solution FreeMore Metallurgy Questions
High purity copper metal is obtained by :...In the electrolysis of molten alumina during the manufacture of aluminium :...Which of the following reaction(s) occur during calcination?...Railway wagon axles are made by heating iron rods embedded in charcoal powder. This process is known as:...The major role of fluorspar (CaF2) which is added in small quantities in the electrolytic reduction of alumina dissolved...