MetallurgyHard
Question
Consider the following isolation / purification processes.
(I) Heating impure metal with I2 at 150 - 200oC and passing the resulting volatile iodide on hot tungsten filament at 1400oC to get the pure metal.
(II) Heating the sulphide ore in air until a part is converted to oxide and then further heating in the absence of air to let the oxide react with unchanged metal sulphide to get the metal.
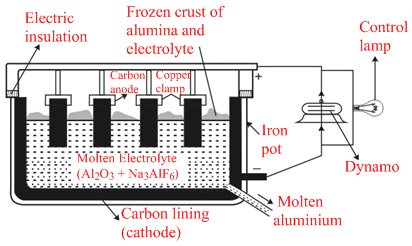
(III) Electrolysis of the molten electrolyte containing metal oxide and cryolite or florspar to obtain the metal.
The processes used for obtaining aluminium, titanium and lead are respectively :
(I) Heating impure metal with I2 at 150 - 200oC and passing the resulting volatile iodide on hot tungsten filament at 1400oC to get the pure metal.
(II) Heating the sulphide ore in air until a part is converted to oxide and then further heating in the absence of air to let the oxide react with unchanged metal sulphide to get the metal.
(III) Electrolysis of the molten electrolyte containing metal oxide and cryolite or florspar to obtain the metal.
The processes used for obtaining aluminium, titanium and lead are respectively :
Options
A.(I), (II) and (III)
B.(II), (III) and (I)
C.(III), (I) and (II)
D.(II), (I) and (III)
Solution
(I) Ti (s) (impure) + 2I2(g) 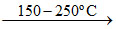 TiI4 (volatile)
TiI4 (volatile) 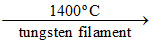 Ti (s) (pure) + 2I2 (g)
Ti (s) (pure) + 2I2 (g)
(II) 2PbS + 3O2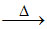 2PbO + 2SO2
2PbO + 2SO2
PbS + 2PbO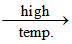 3Pb + SO2
3Pb + SO2
(III) Cathode : Al3+ (melt) + 3e- → Al (l)
Anode : C(s) + O2- (melt) → CO(g) + 2e-
C(s) + 2O2- (melt) → CO2 (g) + 4e-
(II) 2PbS + 3O2
PbS + 2PbO
(III) Cathode : Al3+ (melt) + 3e- → Al (l)
Anode : C(s) + O2- (melt) → CO(g) + 2e-
C(s) + 2O2- (melt) → CO2 (g) + 4e-

Create a free account to view solution
View Solution FreeMore Metallurgy Questions
Which of the following statement(s) is (are) true ?...Which of the following is true for calcination of a metal ore?...Which of the following statement(s) is (are) incorrect ?...Froth flotation is a process of :...Which metal is extracted using a hydrometallurgical process involving complexation?...