MetallurgyHard
Question
In the extraction of aluminium
Process X : employed for red bauxite to remove iron oxide (main impurity)
Process Y : (Serpeck′s process) : used for white bauxite to remove Z (main impurity) then,
Select correct option for the process X and impurity Z.
Process X : employed for red bauxite to remove iron oxide (main impurity)
Process Y : (Serpeck′s process) : used for white bauxite to remove Z (main impurity) then,
Select correct option for the process X and impurity Z.
Options
A.X = Hall and Heroult′s process and Z = SiO2
B.X = Bayer′s process and Z = SiO2
C.X = Serpeck′s process and Y = iron oxide
D.X = Bayer′s process and Y = iron oxide
Solution
(X) Red bauxite contains the impurities of oxides of iron and silicates. In Bayer process, alumina isdissolved by reacting sodium hydroxide solution leaving behind the insoluble oxide of iron.
(Y) White bauxite contains the impurity of silica which is removed by Serpeck′s method.
Al2O3 + N2 + 3C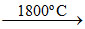 2AlN + 3CO ; SiO2 + 2C
2AlN + 3CO ; SiO2 + 2C 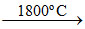 Si ↑+ 2CO↑
Si ↑+ 2CO↑
AlN + 3H2O → Al(OH)3↓ + NH3 ; 2Al(OH)3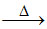 Al2O3 + 3H2O
Al2O3 + 3H2O
(Y) White bauxite contains the impurity of silica which is removed by Serpeck′s method.
Al2O3 + N2 + 3C
AlN + 3H2O → Al(OH)3↓ + NH3 ; 2Al(OH)3
Create a free account to view solution
View Solution FreeMore Metallurgy Questions
Which of the following pair consists of ore of the same metal?...Which of the following is(are) regarded as iron ores?...In which of the following pairs, both the minerals are oxides?...The slag consists of molten impurities, generally, in the form of :...Which mineral has been named incorrectly ?...