General Organic ChemistryHard
Question
What is the decreasing order of strength of the bases OH-, NH2-, H - C ≡ C- and CH3 - CH2- ?
Options
A.CH3 - CH2- > NH2- > H - C ≡ C- > OH-
B.H - C ≡ C- > CH3 - CH2- > NH2- > OH-
C.OH- > NH2- > H - C ≡ C- > CH3 - CH2-
D.NH2- > H - C ≡ C-> OH- > CH3 - CH2-
Solution
Conjugate base of dtrong acid weaker while conjugate base of weak acid is stronger because the acidic strength folloes the order. Thus
H - OH > HC ≡ CH > NH3 > CH3 - CH3
Order of basic strength :
CH3CH2- >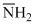 > HC ≡ C- < OH-
> HC ≡ C- < OH-
H - OH > HC ≡ CH > NH3 > CH3 - CH3
Order of basic strength :
CH3CH2- >
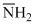 > HC ≡ C- < OH-
> HC ≡ C- < OH- Create a free account to view solution
View Solution FreeMore General Organic Chemistry Questions
Which among the following compounds behave both as an electrophile as well as a nucleophile: (a) CH2 = CH2 (b) CH2 = CH ...Which one of the nitrogen containing compounds is an electrophile :...The order of stability of the following carbanion is : (I) (II) (III) (IV)...The correct order of increasing basicity is -...The correct statement regarding electrophile is :-...