General Organic ChemistryHard
Question
The molecule (s) that will have dipole moment is / are :
Options
A.2, 2 dimethyl propane
B.trans -2-pentene
C.cis-3-hexene
D.2,2,3,3 -tetramethyl butane
Solution
(a) 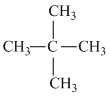
(μ = 0, due symmetrical structure.)
(b)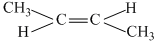
μ > 0, unsymmetrical molecule
(c)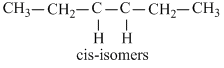
μ > 0 μresultant will not be zero in cis-siomers
(d)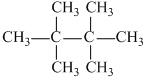
(μ = 0, symmetrical molecule.)

(μ = 0, due symmetrical structure.)
(b)
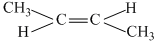
μ > 0, unsymmetrical molecule
(c)
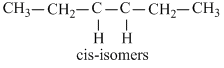
μ > 0 μresultant will not be zero in cis-siomers
(d)

(μ = 0, symmetrical molecule.)
Create a free account to view solution
View Solution FreeMore General Organic Chemistry Questions
The enolic form of acetone contais :...Which of the following carbocations is expected to be most stable ?...In the anion HCOO−, the two carbon-oxygen bonds are found to be equal length. What is the reason for it −...Increasing order of the stability is CH3CH = CH2 CH3CH2CH = CH2, I II CHCH = CH2 (CH3)3CCH = CH2 III IV...The ortho/para directing group among the following is :...