General Organic ChemistryHard
Question
The compound in which C uses its sp3 - hybrid orbitals for bond formation is :
Options
A.HCOOH
B.(H2N)2CO
C.(CH3)3COH
D.CH3CHO
Solution
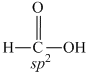 NH2 - CONH2
NH2 - CONH2sp2
(CH3)3 - OH CH3CHO
sp3 sp3
Create a free account to view solution
View Solution FreeMore General Organic Chemistry Questions
IUPAC name of...Among the following, the compound that can be most redily sulphonated is :...In following acid, the correct increasing order of acidic strength acidHCOOH, CH3COOH, C6H5COOH I II III...The number of sigma electrons in toluene are...The order of stability of the following carbocations : is :-...