MetallurgyHard
Question
Which of the following reactions represents the self-reduction process?
Options
A.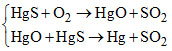
B.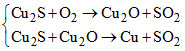
C.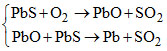
D.All of these
Solution
Sulphide ore of Hg, Cu, Pb are heated in air, a part of these is changed in to oxides or sulphate that then react with the remaining part of the sulphide ore to give its metal and SO2. This is called self reduction, auto reduction or air reduction method.
Create a free account to view solution
View Solution FreeMore Metallurgy Questions
Which of the following compounds is not colored yellow?...Si and Ge used for semiconductors are required to be of high purity and hence purified by...High purity copper metal is obtained by :...In the extraction of copper from its sulphide ore, the metal is formed by reduction of Cu2O with :...Which mineral has been named incorrectly ?...