MetallurgyHard
Question
On the basis of ellingham diagram which of the following is/are correct.
Options
A.Entropy change for all metal oxides is roughly same.
B.Below the boiling point, ′TᐃS′ factor is same irrespective of metal.
C.Above ᐃG = 0 line, oxide decomposes into metal & oxygen.
D.If randomness increases the slope increases
Solution
(A) 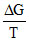 = - ᐃS,
= - ᐃS, 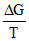 is slope in Ellingham diagram. Which is same below the boiling point.
is slope in Ellingham diagram. Which is same below the boiling point.
(B) Below the boiling point slope is same as factor TᐃS is same.
(C) Above ᐃG = 0 line free energy becomes positive so oxide decomposes.
(D) Random increases i.e. ᐃS increases, so slope also increases.
(B) Below the boiling point slope is same as factor TᐃS is same.
(C) Above ᐃG = 0 line free energy becomes positive so oxide decomposes.
(D) Random increases i.e. ᐃS increases, so slope also increases.
Create a free account to view solution
View Solution FreeMore Metallurgy Questions
In the aluminothermite process, aluminium acts as :...Which of the following employ(s) thermal decomposition of volatile iodide compounds?...For extraction of sodium from NaCl, the electrolytic mixture NaCl + KCl + CaCl2 is used. During extraction process, only...Cryolite is :...Auto reduction process is used in extraction of...