General Organic ChemistryHard
Question
Amongst the following , the most basic compound is :
Options
A.benzylamine
B.aniline
C.acetanilide
D.p-nitroaniline
Solution
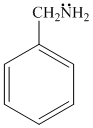
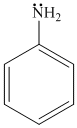
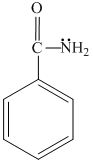
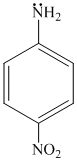
(a) (b) (c) (d)
In (b) and (d) ,lp of
 gp. enters in resonance with benzene ring, hence are not readily available for protonation. In (c) , it enters in resonance with
gp. enters in resonance with benzene ring, hence are not readily available for protonation. In (c) , it enters in resonance with 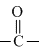 gp.(lp - π bond interaction)
gp.(lp - π bond interaction)In (a), lp of -
 can′t enter in resonance and so is available for protonation most easily,i.e., why benzylamine is most basic .
can′t enter in resonance and so is available for protonation most easily,i.e., why benzylamine is most basic .Create a free account to view solution
View Solution FreeMore General Organic Chemistry Questions
Give the correct order of increasing acidity of the following compounds -(I) OH (II) OH(III) COOH (IV) C − CH...Consider the following compoundWhich of the above compounds reacts with NaHCO3 giving CO2...Match the reactions in column I with appropriate types of steps/reactive intermediate involved in these reactions as giv...Correct order of leaving group tendency -...Among the following, l-serine is :...