MetallurgyHard
Question
Consider the following statements :
S1 : In electrolytic refining, the impurities from the blister copper deposits anode mud which contains antimony, selenium, tellurium, silver, gold and platinum. (From copper pyrites)
S2 : In Serpeck’s process silica is removed by heating the bauxite to 1800oC with coke in a current of N2 .
S3 : Chalcocite and azurite are ores of copper.
S4 : The tin is obtained by the carbon reduction of black tin.
and arrange in the order of true/false
S1 : In electrolytic refining, the impurities from the blister copper deposits anode mud which contains antimony, selenium, tellurium, silver, gold and platinum. (From copper pyrites)
S2 : In Serpeck’s process silica is removed by heating the bauxite to 1800oC with coke in a current of N2 .
S3 : Chalcocite and azurite are ores of copper.
S4 : The tin is obtained by the carbon reduction of black tin.
and arrange in the order of true/false
Options
A.T F T T
B.F T F F
C.F F T T
D.T T T T
Solution
S1 : The mud obtained below the anode contains unreactive Sb, Se, Te, Ag, Au and Pt.
S2 : SiO2 + 2C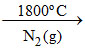 Si ↑+ 2CO↑ (Serpeck′s method)
Si ↑+ 2CO↑ (Serpeck′s method)
At this temperature silicon is volatile.
S3 : Chalco pyrite is CuFeS2 and azurite is 2CuCO3.Cu(OH)2.
S4 : Purified cassiterite ore containg 60 -70% SnO2 is called black tin.
SnO2 (black tin) + 2C → Sn + 2CO (carbon reduction).
S2 : SiO2 + 2C
At this temperature silicon is volatile.
S3 : Chalco pyrite is CuFeS2 and azurite is 2CuCO3.Cu(OH)2.
S4 : Purified cassiterite ore containg 60 -70% SnO2 is called black tin.
SnO2 (black tin) + 2C → Sn + 2CO (carbon reduction).
Create a free account to view solution
View Solution FreeMore Metallurgy Questions
Froth floatation :...Zone refining is based on the principle of...The chief reaction(s) occurring in blast furnace during extraction of iron from haematite is(are)...During the electrolysis of fused carnallite, MgCl2 is decomposed to liberate Mg at cathode and not KCl to liberate the K...In the extraction of copper from its sulphide ore the metal is formed by the reduction of Cu2O with:...