General Organic ChemistryHard
Question
The bond between carbon atom (1) and carbon atom (2) in compound
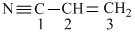
involves the hybridisation as :
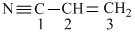
involves the hybridisation as :
Options
A.sp2 and sp2
B.sp3 and sp
C.sp and sp2
D.sp and sp
Solution
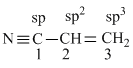
C1 - C2 bond involves sp - sp2 hybridization.
Create a free account to view solution
View Solution FreeMore General Organic Chemistry Questions
Arrange the following alkenes in decreasing order of stability.I. II. III. IV. Choose the correct answer from the option...Carbocations may be stabilised by -...Which is not aromatic compound ?...The order of stability of the following carbocations : is :-...Which is the correct order of increasing basicity ?...