MetallurgyHard
Question
Of the following reduction processes, the correct process(es) is/are :
Options
A.B2O3 + Al 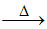 B.
B.
B.Cr2O3 + 2Al 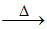 Cr.
Cr.
C.TiCl4 + Mg 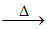 Ti.
Ti.
D.PbS + PbO 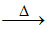 Pb.
Pb.
Solution
β(A) B2O3 + 2Al 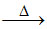 2B + Al2O3 (aluminothermic process-extraction of boron)
2B + Al2O3 (aluminothermic process-extraction of boron)
(B) Cr2O3 + Al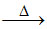 2Cr + Al2O3 (extraction of chromium)
2Cr + Al2O3 (extraction of chromium)
(C) TiCl4 + 2Mg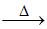 Ti + 2MgCl2 (Kroll process-extraction of titanium)
Ti + 2MgCl2 (Kroll process-extraction of titanium)
(D) PbS + 2PbO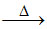 3Pb + SO2 (extraction of lead)
3Pb + SO2 (extraction of lead)
(B) Cr2O3 + Al
(C) TiCl4 + 2Mg
(D) PbS + 2PbO
Create a free account to view solution
View Solution FreeMore Metallurgy Questions
Mac Arthur process is used for the extraction of :...Gravity separation method is based upon :...Which one of the following metals cannot be extracted by carbon reduction ?...Addition of high proportions of manganese makes steel useful in making rails of railroads, because manganese...High purity copper metal is obtained by :...