HydrocarbonsHard
Question
Consider the following reaction :
(i) CH3 - CH2 - CH3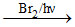 P1
P1
(ii) CH3 - CH2 - CH3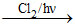 P2
P2
Choose the correct alternative
(i) CH3 - CH2 - CH3
(ii) CH3 - CH2 - CH3
Choose the correct alternative
Options
A.P1 is 2-bromopropane while P2 is 1- chloropropane
B.P1 is 2-bromopropane while P2 is 2- chloropropane
C.P1 is 1-bromopropane mainly and P2 is a mixture of 1-chloropropane and 2-chloropropane
D.P1 is 2-Bromopropane mainly and P2 is a mixture of 1-chloropropane and 2-chloropropane
Solution
Bromination is more selective than chlorination,so forms most stable products only.
Create a free account to view solution
View Solution Free