MetallurgyHard
Question
Leaching of Ag2S is carried out by heating it with a dilute solution of :
Options
A.NaCN only
B.HCI
C.NaOH
D.NaCN in presence of O2
Solution
Ag2S (conc. ore) + 2NaCN 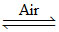 2AgCN + Na2S.
2AgCN + Na2S.
Ag2S and AgCN are in equilibrium so Na2S is oxidised by air in to Na2SO4. Hence equilibrium shifts towards right side.
4Na2S + 5O2 + 2H2O → 2Na2SO4 + 4NaOH + 2S
AgCN + NaCN → Na[Ag(CN)2] (soluble complex)
2Na[Ag(CN)2] + Zn (dust) → 2Ag+ ↓ Na2[Zn(CN)4].
Ag2S and AgCN are in equilibrium so Na2S is oxidised by air in to Na2SO4. Hence equilibrium shifts towards right side.
4Na2S + 5O2 + 2H2O → 2Na2SO4 + 4NaOH + 2S
AgCN + NaCN → Na[Ag(CN)2] (soluble complex)
2Na[Ag(CN)2] + Zn (dust) → 2Ag+ ↓ Na2[Zn(CN)4].
Create a free account to view solution
View Solution FreeMore Metallurgy Questions
The correct statements are:...During electrolytic reduction of alumina, two auxiliary electrolytes X and Y are added to increase the electrical conduc...NaCl and CaCl2 are added to fused MgCl2 in the electrolysis of MgCl2 since :...Aluminium is extracted from alumina (Al2O3) by electrolysis of a molten mixture of :...Slag is a product of :...