MetallurgyHard
Question
The following flow diagram represents the extraction of magnesium from sea water.
Sea water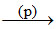 Mg(OH)2
Mg(OH)2 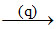 MgCl2 + H2O
MgCl2 + H2O
↓ crystallisation
Magnesium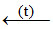 Anhydrous MgCl2
Anhydrous MgCl2 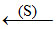 (r)
(r)
Which of the following option describes the correct reactants, products and reaction conditions ?
Sea water
↓ crystallisation
Magnesium
Which of the following option describes the correct reactants, products and reaction conditions ?
Options
A.(p) → Milk of magnesia
(q) → HCl(aq.)
(r) → MgCl2.2H2O
(s) → Heating in limited supply of air
(t) → Reduction by coke.
(q) → HCl(aq.)
(r) → MgCl2.2H2O
(s) → Heating in limited supply of air
(t) → Reduction by coke.
B.(p) → Lime water
(q) → HCl(aq.)
(r) → MgCl2
(s) → Heating in absence of air
(t) → Electrolytic reduction in molten state.
(q) → HCl(aq.)
(r) → MgCl2
(s) → Heating in absence of air
(t) → Electrolytic reduction in molten state.
C.(p) → Milk of lime
(q) → HCl(aq.)
(r) → MgCl2.6H2O
(s) → Heating in presence of dry HCl
(t) → Electrolytic reduction in molten state.
(q) → HCl(aq.)
(r) → MgCl2.6H2O
(s) → Heating in presence of dry HCl
(t) → Electrolytic reduction in molten state.
D.(p) → Milk of lime
(q) → HCl(aq.)
(r) → MgCl2.6H2O
(s) → Heating in presence of dry HCl
(t) → Electrolysis of aqueous solution
(q) → HCl(aq.)
(r) → MgCl2.6H2O
(s) → Heating in presence of dry HCl
(t) → Electrolysis of aqueous solution
Solution
MgCl2 + Ca(OH)2(p) → Mg(OH)2 ↓ + HCl(q) → MgCl2 + H2O 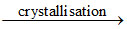 MgCl2.6H2O (r)
MgCl2.6H2O (r)


Create a free account to view solution
View Solution FreeMore Metallurgy Questions
An non-magnetic ore containing the impurity of FeCr2O4 is concentrated by...Which of the following statements is correct regarding the slag obtained during the extraction of a metal like copper or...Chemical leaching is useful in the concentration of:...The process of removing lighter gangue particles by washing in a current of water is called :...The metal which is obtained from both sea-water and ores from the earth′s solid crust is :...